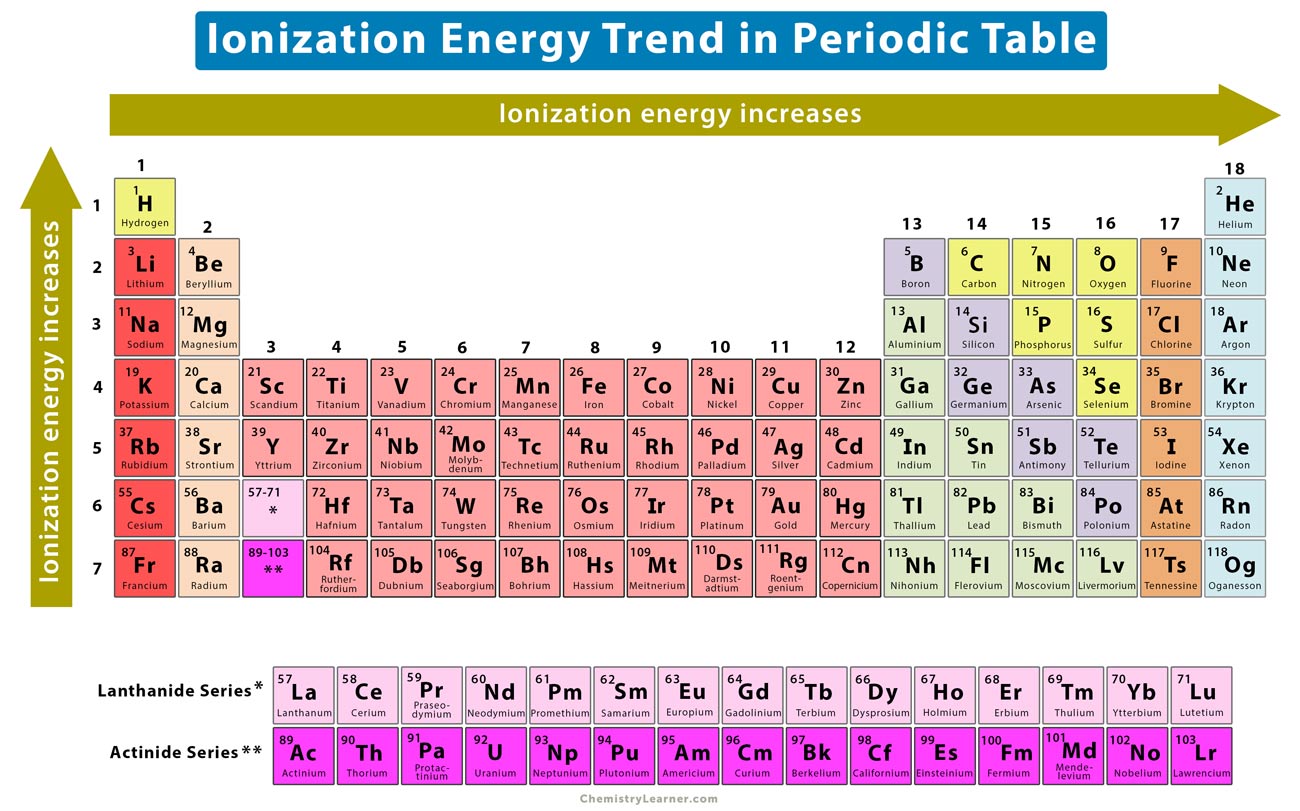
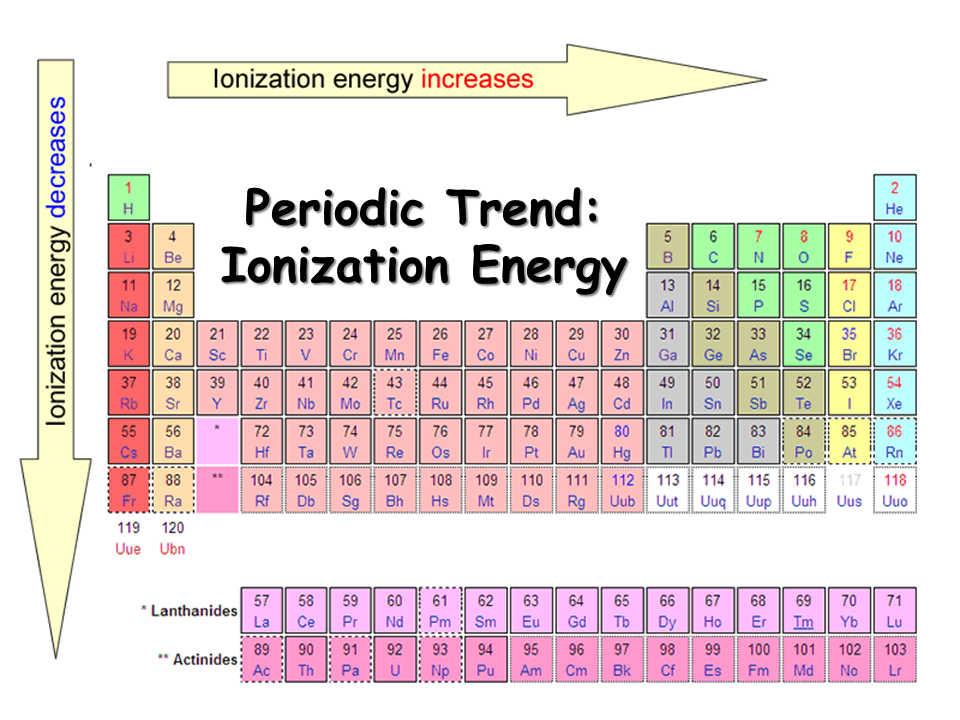
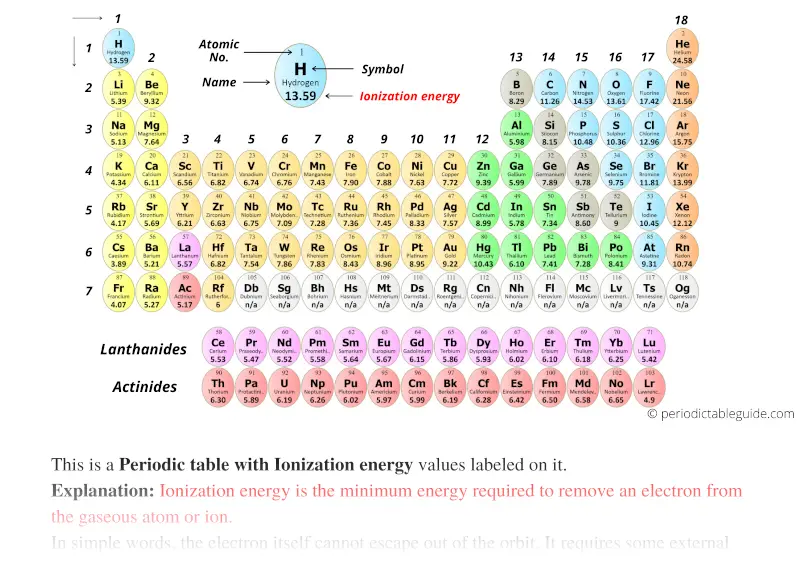
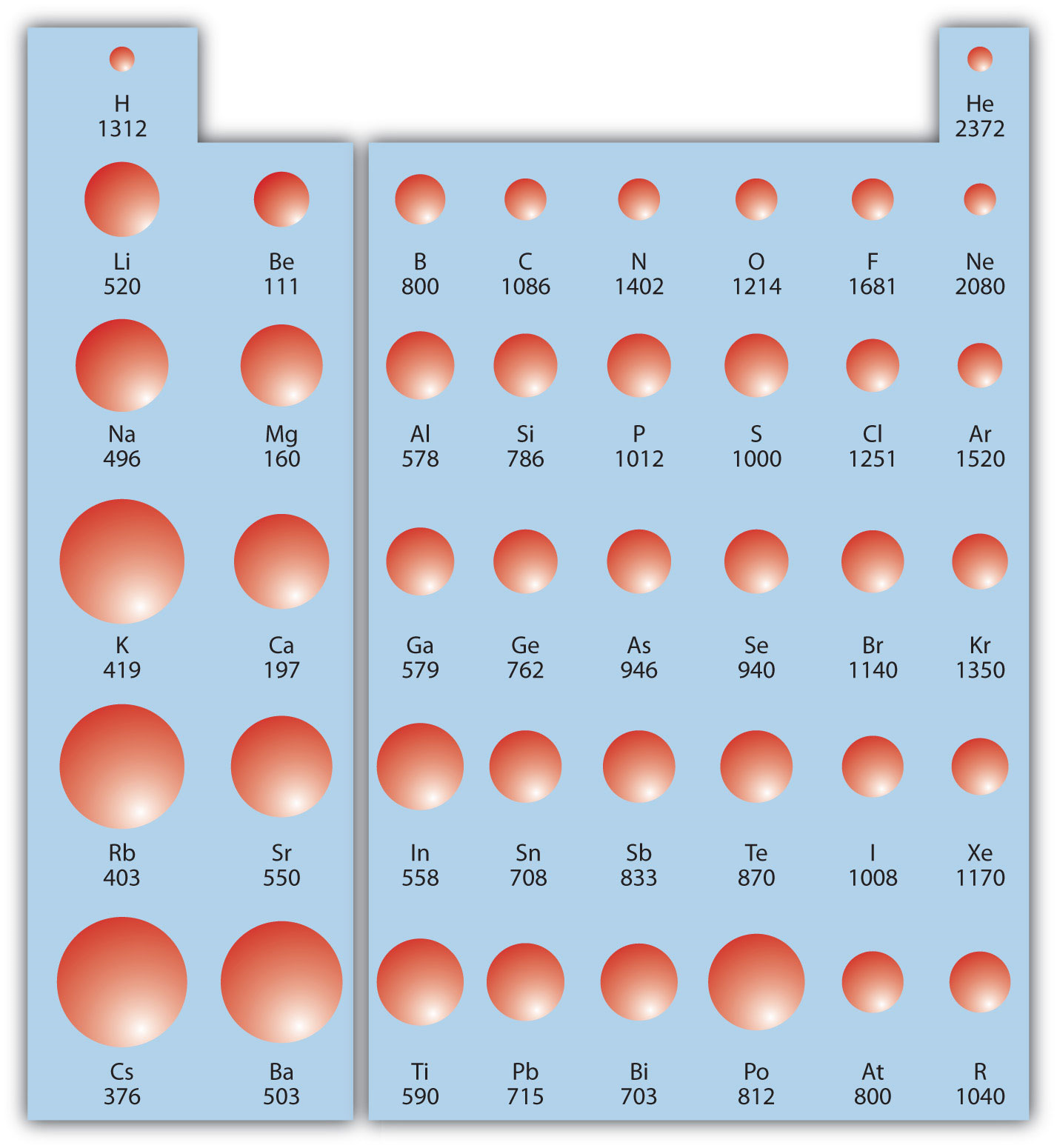
Ionization Energy Chart - The measurement is performed in the gas phase on single atoms. 1 ev / atom = 96.49 kj / mol. Web ionization energy is the energy required to remove an electron from a neutral atom in its gaseous phase. The first molar ionization energy applies to the neutral atoms. Web the ionization energy of atoms, denoted e i, is measured by finding the minimal energy of light quanta or electrons accelerated to a known energy that will kick out the least bound atomic electrons. Web molar ionization energies of the elements. Ionization energy is always positive. It measures the capability of an atom to lose an electron during a chemical reaction. Alkali metals alkaline earth metals. First ionization energy, second ionization energy as well as third ionization energy of the elements are given in this chart.
Among the Following Which Element Has the Lowest Ionization Energy
First ionization energy, second ionization energy as well as third ionization energy of the elements are given in this chart. Web explore how ionization energy.
What Is Ionization Energy? Definition and Trend
Web ionization energy is the minimum energy required to remove a loosely bound electron of an atom or molecule in the gaseous state. 1 ev.
Ionization Enthalpy NEET Lab
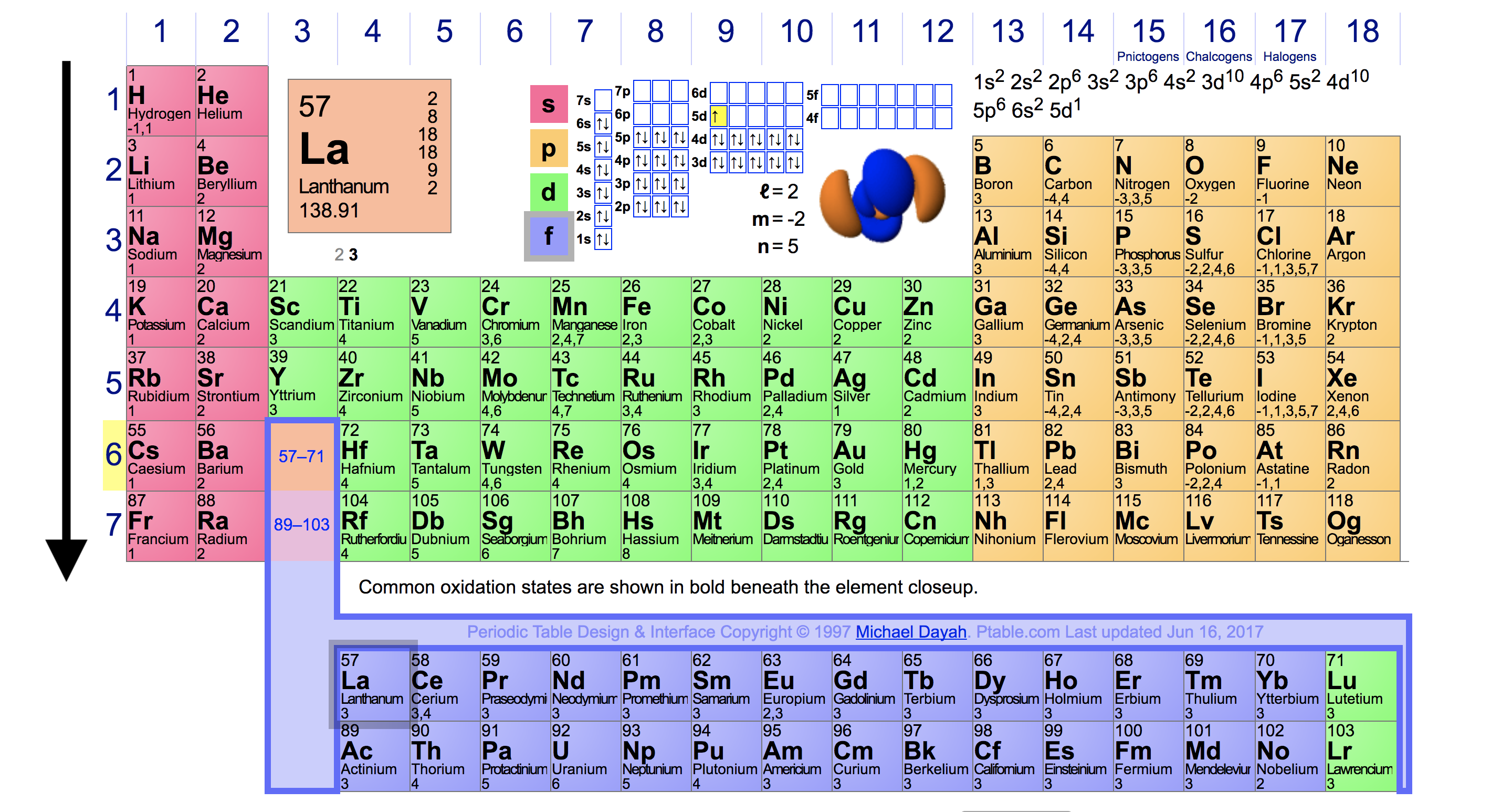
Web complete and detailed technical data about the element $$$elementname$$$ in the periodic table. Web explore how ionization energy changes with atomic number in the.
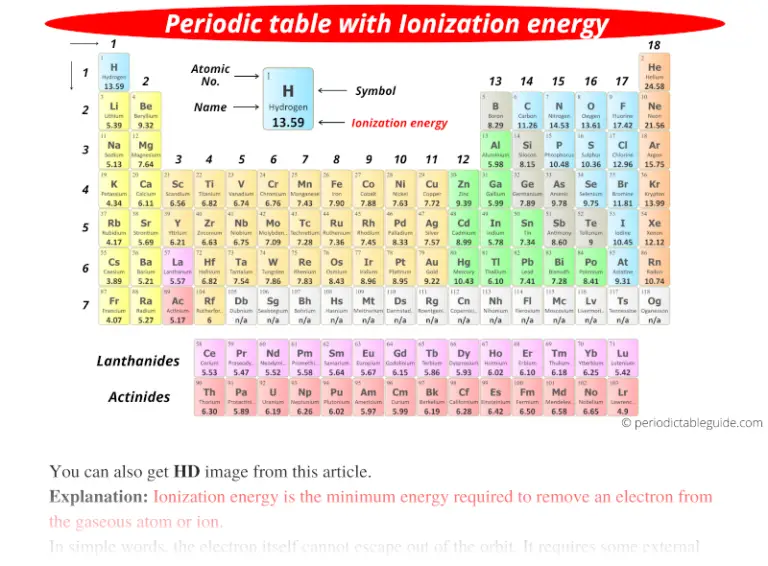
Periodic table with Ionization Energy Values (Labeled Image)
Alkali metals alkaline earth metals. Web typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): The first molar ionization energy applies to.
Periodic Trends in Ionization Energy Chemistry Socratic
This is the energy per mole necessary to remove electrons from gaseous atoms or atomic ions. It measures the capability of an atom to lose.
Periodic Trends in Ionization Energy CK12 Foundation
Web explore how ionization energy changes with atomic number in the periodic table of elements via interactive plots. Web complete and detailed technical data about.
6.4 Ionization Energy Chemistry LibreTexts
Web the ionization energy of atoms, denoted e i, is measured by finding the minimal energy of light quanta or electrons accelerated to a known.
9.9 Periodic Trends Atomic Size, Ionization Energy, and Metallic
Ionization energy is always positive. The measurement is performed in the gas phase on single atoms. Web typical units for ionization energies are kilojoules/mole (kj/mol).
The Parts of the Periodic Table
Web complete and detailed technical data about the element $$$elementname$$$ in the periodic table. Web the periodic table of the elements (with ionization energies) 1..
The Ionization Energy Of The Elements Within A Period.
This is the energy per mole necessary to remove electrons from gaseous atoms or atomic ions. Web ionization energy chart of all the elements is given below. Web complete and detailed technical data about the element $$$elementname$$$ in the periodic table. The first molar ionization energy applies to the neutral atoms.
Web The Periodic Table Of The Elements (With Ionization Energies) 1.
Alkali metals alkaline earth metals. Web for each atom, the column marked 1 is the first ionization energy to ionize the neutral atom, the column marked 2 is the second ionization energy to remove a second electron from the +1 ion, the column marked 3 is the third ionization energy to remove a third electron from the +2 ion, and so on. Web explore how ionization energy changes with atomic number in the periodic table of elements via interactive plots. 1 ev / atom = 96.49 kj / mol.
If An Atom Possesses More Than One Electron, The Amount Of Energy Needed To Remove Successive Electrons Increases Steadily.
Ionization energy is always positive. Web ionization energy is the energy required to remove an electron from a neutral atom in its gaseous phase. First ionization energy, second ionization energy as well as third ionization energy of the elements are given in this chart. Web typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev):
It Measures The Capability Of An Atom To Lose An Electron During A Chemical Reaction.
The measurement is performed in the gas phase on single atoms. Web ionization energy is the minimum energy required to remove a loosely bound electron of an atom or molecule in the gaseous state. These tables list values of molar ionization energies, measured in kj⋅mol −1. Web molar ionization energies of the elements.