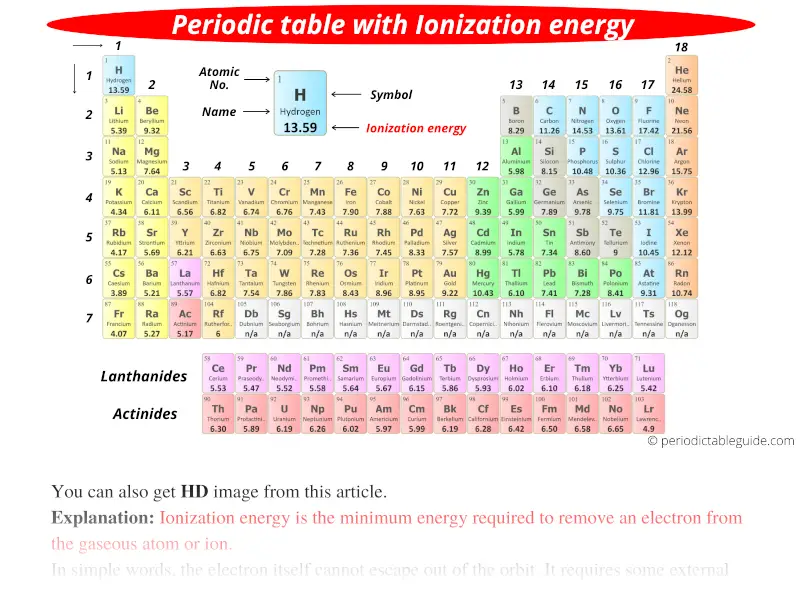
Ionic Energy Chart - Hundreds of chemical reactions in your body involve magnesium. On the periodic table, first. Web ionization energy chart of all the elements is given below. Web ionization energy, in chemistry and physics, the amount of energy required to remove an electron from an isolated atom or molecule. The energy required to remove an electron is the ionization energy. Web for each atom, the column marked 1 is the first ionization energy to ionize the neutral atom, the column marked 2 is the second ionization energy to remove a second electron. Your body needs it for muscle. The energy required to remove the outermost electron from an atom or a positive ion in its ground level. The table lists only the first ie in ev units. Web chemists define the ionization energy (\(i\)) of an element as the amount of energy needed to remove an electron from the gaseous atom \(e\) in its ground state.
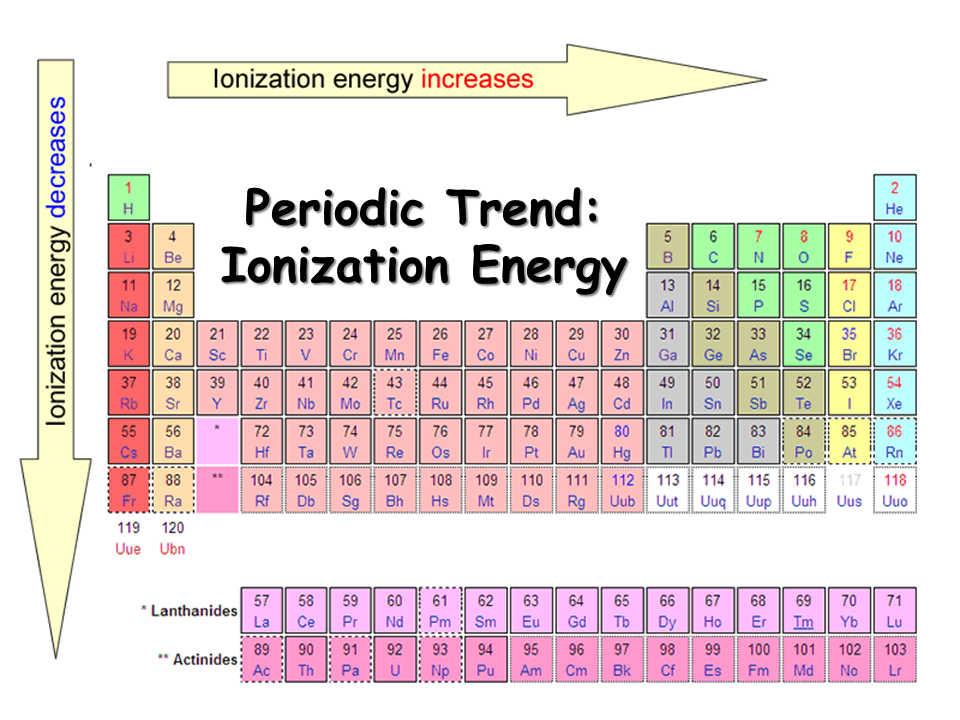
Periodic table with Ionization Energy Values (Labeled Image)
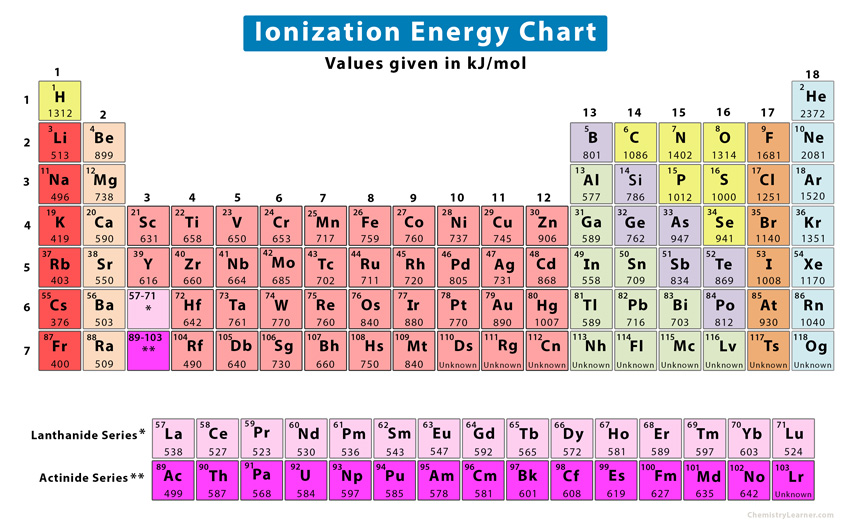
The table lists only the first ie in ev units. The first ionization energy, second. The energy required to remove an electron is the ionization.
Example
Web magnesium is one of the most abundant minerals in your body. Web when electrons are removed in succession from an element, the transition from.
Periodic Table Ionization Energy Labeled
Another is when each of 3 p orbitals have one. The stronger an electron is bound to an atom the more. Ionization energy is always.
Periodic Table Ionization Energy Chart
The stronger an electron is bound to an atom the more. The energy required to remove the outermost electron from an atom or a positive.
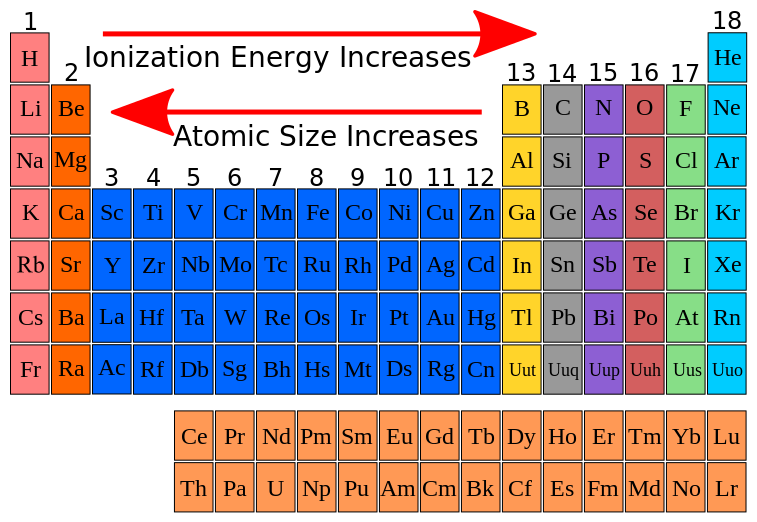
9.9 Periodic Trends Atomic Size, Ionization Energy, and Metallic
Web ionization energy, in chemistry and physics, the amount of energy required to remove an electron from an isolated atom or molecule. The most common.
Ionization Energy Definition, Chart & Periodic Table Trend
Web for each atom, the column marked 1 is the first ionization energy to ionize the neutral atom, the column marked 2 is the second.
What Is Ionization Energy? Definition and Trend
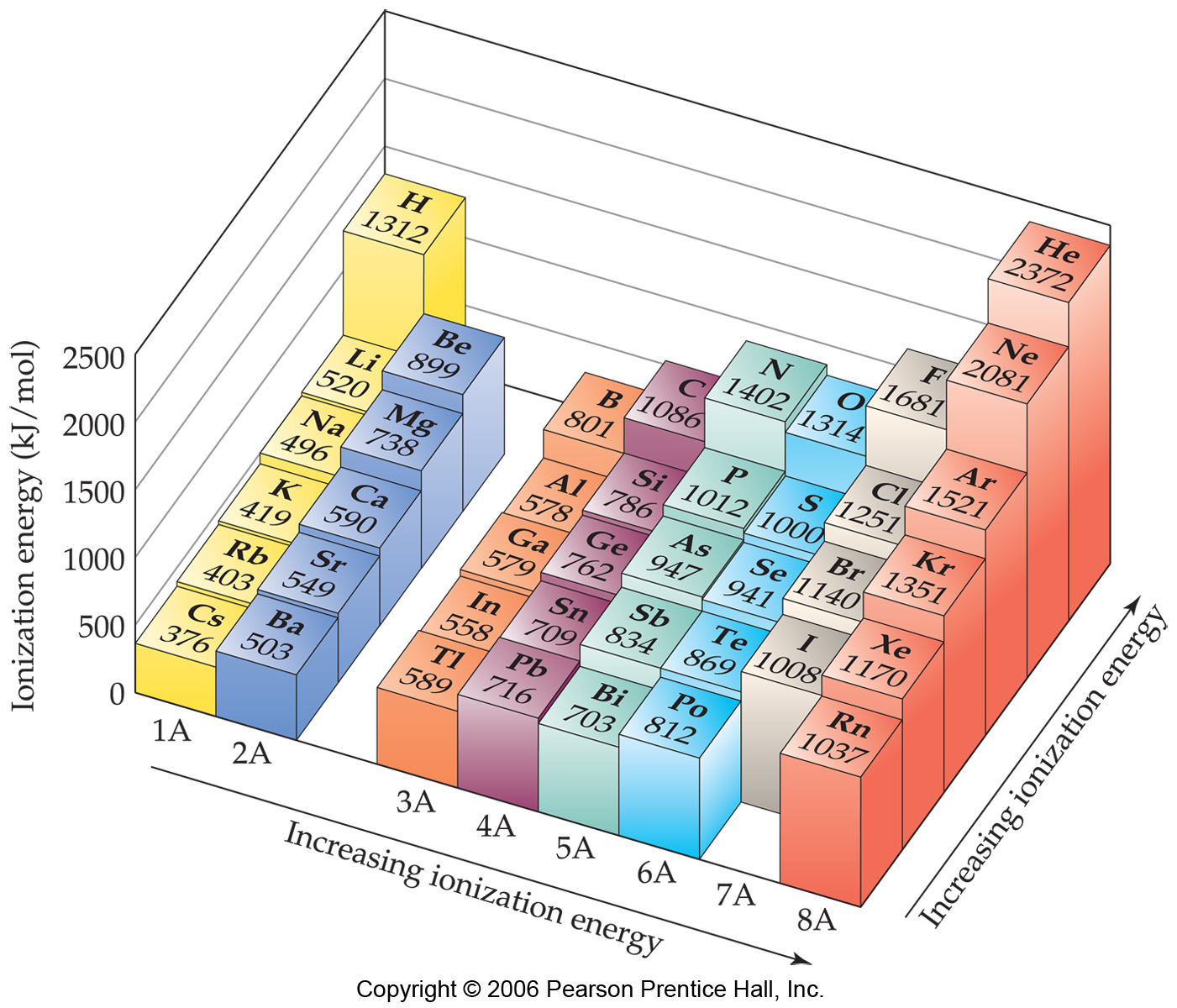
As you can see on the graph, the noble gases have the highest ionization energies, and the alkali metals have the lowest ionization. Your body.
Ionization Enthalpy NEET Lab
Web when electrons are removed in succession from an element, the transition from removing valence electrons to removing core electrons results in a large jump.
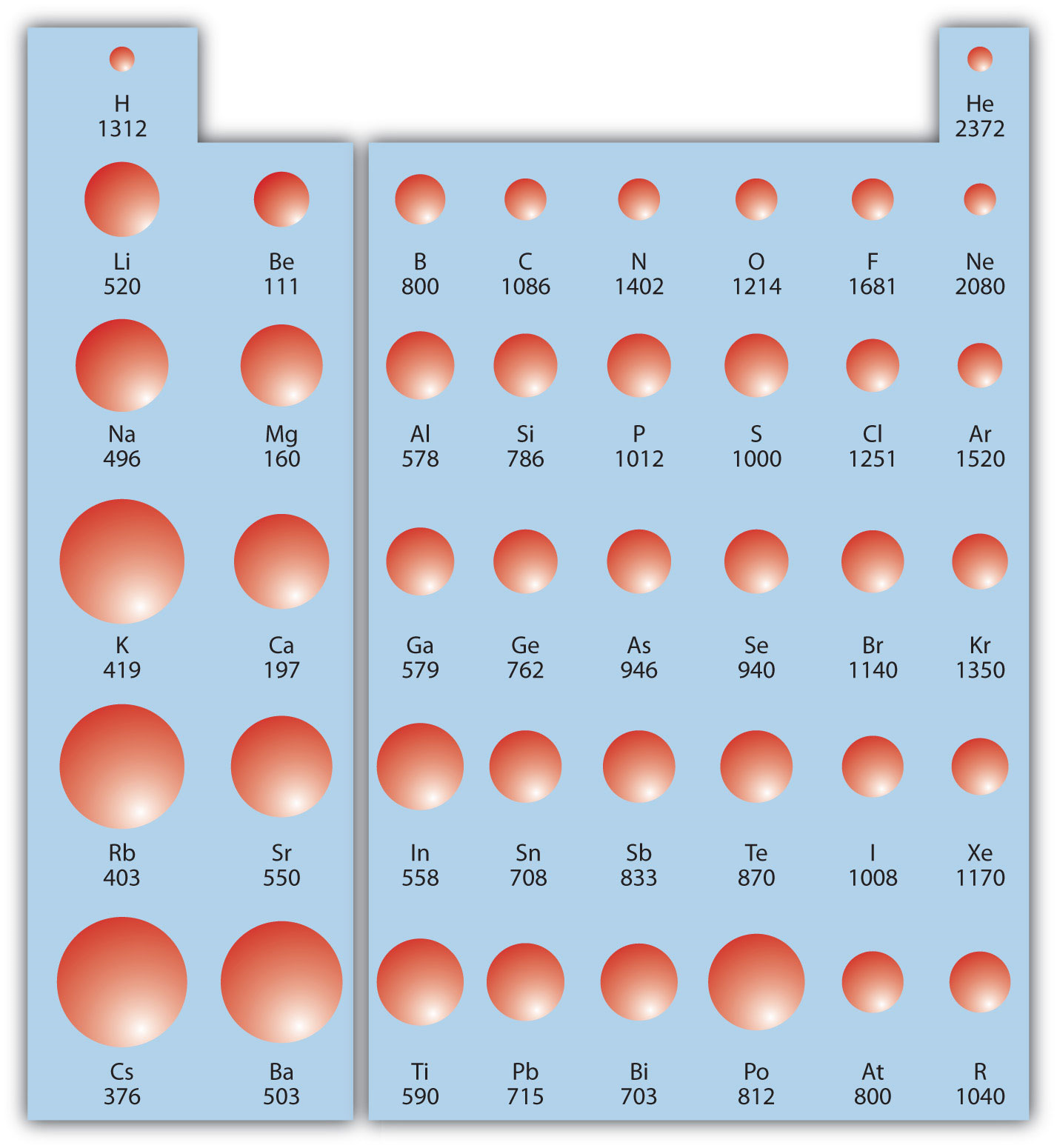
FileIonization energy atomic size.svg Wikimedia Commons
On the periodic table, first. Web ionization is the process of removing an electron from a neutral atom (or compound). Web looking at the graph.
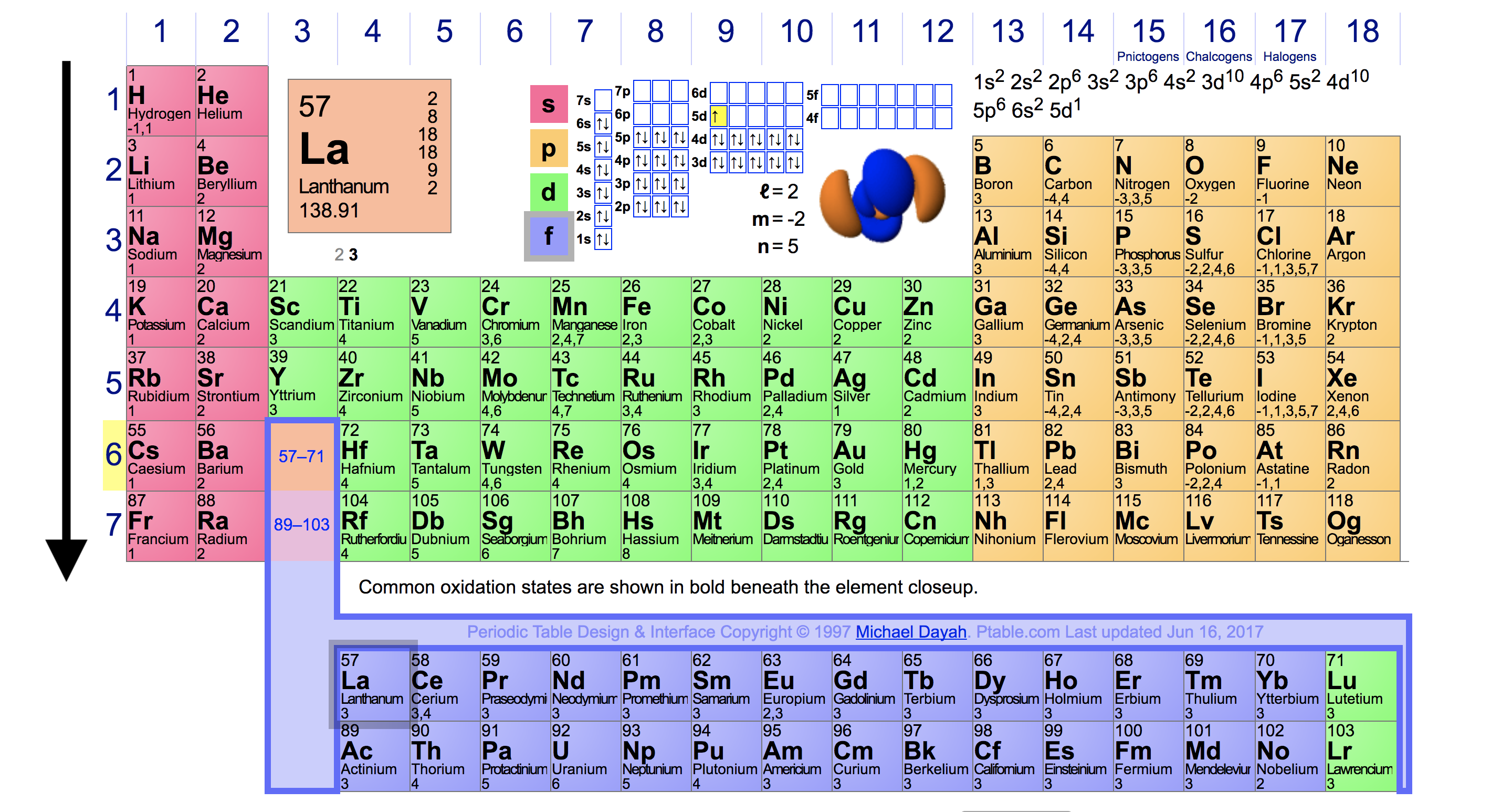
Web Molar Ionization Energies Of The Elements.
The energy required to remove the outermost electron from an atom or a positive ion in its ground level. Web chemists define the ionization energy (\(i\)) of an element as the amount of energy needed to remove an electron from the gaseous atom \(e\) in its ground state. Ionization energy is always positive. Web ionization energy is the minimum energy required to remove an electron from an atom or ion in the gas phase.
Web The Ionization Energy Of Atoms, Denoted E I, Is Measured By Finding The Minimal Energy Of Light Quanta Or Electrons Accelerated To A Known Energy That Will Kick Out The Least Bound.
Web ionization energy is a measure of the energy needed to pull a particular electron away from the attraction of the nucleus. Web an element's first ionization energy is the energy required to remove the outermost, or least bound, electron from a neutral atom of the element. Hundreds of chemical reactions in your body involve magnesium. Web for each atom, the column marked 1 is the first ionization energy to ionize the neutral atom, the column marked 2 is the second ionization energy to remove a second electron.
Web When Electrons Are Removed In Succession From An Element, The Transition From Removing Valence Electrons To Removing Core Electrons Results In A Large Jump In Ionization.
This is the energy per mole necessary to remove. The energy required to remove an electron is the ionization energy. Web ionization energy, in chemistry and physics, the amount of energy required to remove an electron from an isolated atom or molecule. Your body needs it for muscle.
As You Can See On The Graph, The Noble Gases Have The Highest Ionization Energies, And The Alkali Metals Have The Lowest Ionization.
The ionization energy associated with. The stronger an electron is bound to an atom the more. These tables list values of molar ionization energies, measured in kj⋅mol −1. Web ionization energy is the quantity of energy that an isolated, gaseous atom in the ground electronic state must absorb to discharge an electron, resulting in a cation.